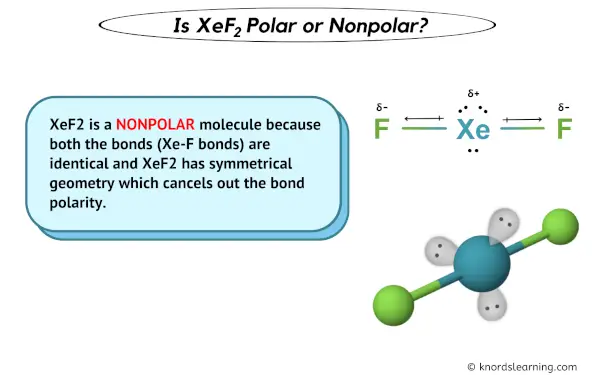
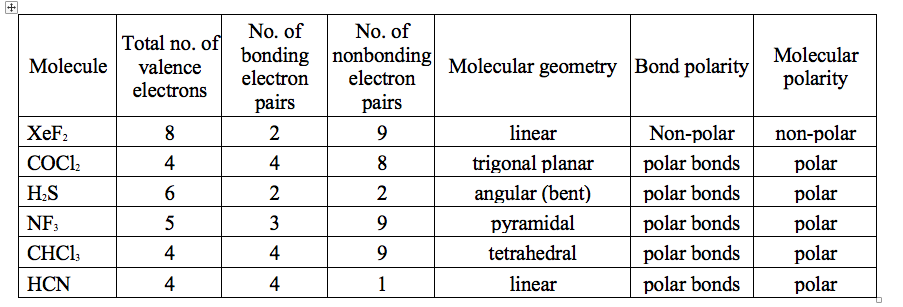
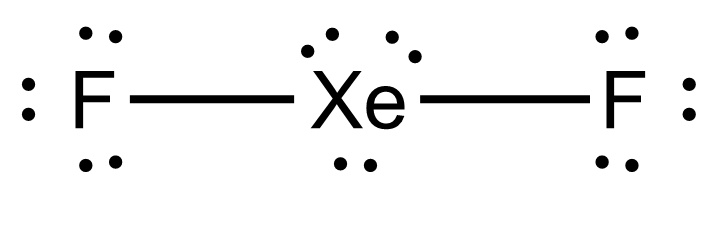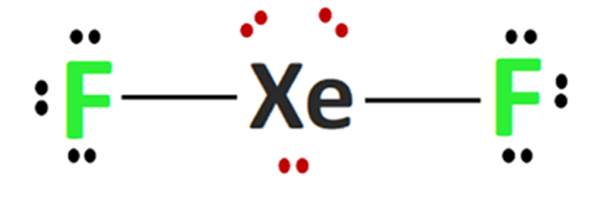What is the shape of the XeF2 molecule, and the total number of the lone pair present on XE in a XeF2 molecule? - Quora

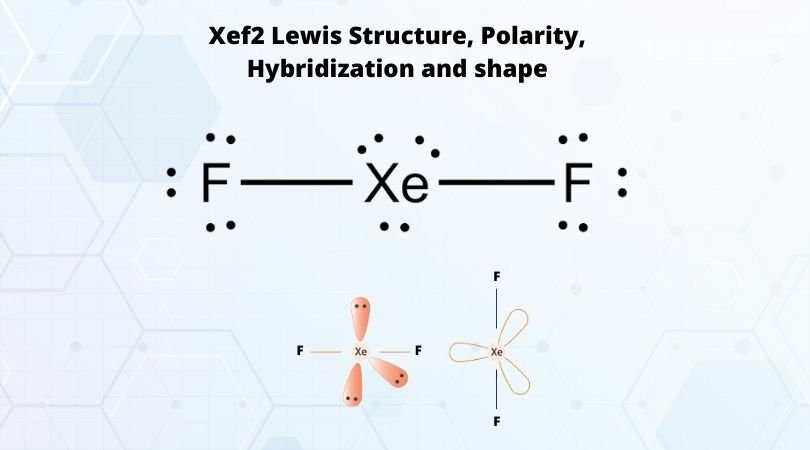
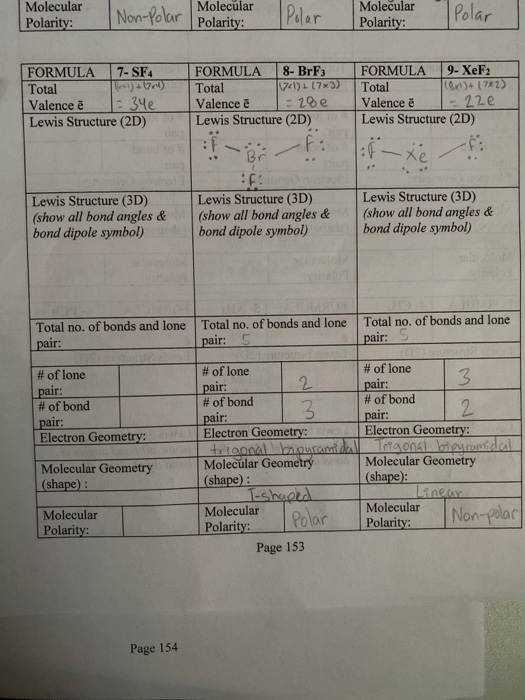
Draw the Lewis structure for XeF2 and provide the following information. a. electron-pair geometry b. molecular geometry c. hybridization of the central atom d. dipole moment | Homework.Study.com