Is Aspirin Polar or Nonpolar? – (Polarity of Aspirin) | Molecular geometry, Molecular shapes, Organic molecules

Aspirin. An ab Initio Quantum-Mechanical Study of Conformational Preferences and of Neighboring Group Interactions | The Journal of Organic Chemistry
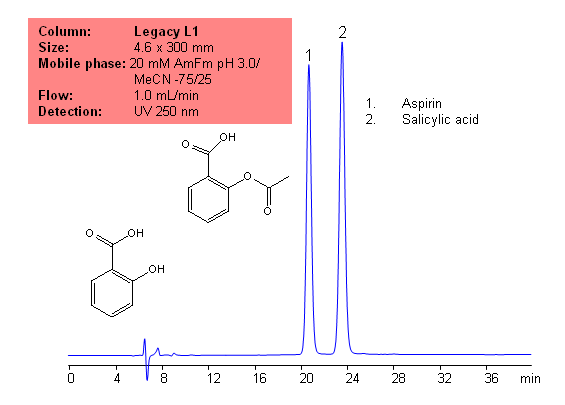
USP Methods for the Analysis of Aspirin (Acetylsalicylic acid (ASA)) Using Legacy L1 Column | SIELC Technologies

Following is a molecular model of aspirin (acetylsalicylic acid). Identify the hybridization of the orbitals on each carbon atom in aspirin, and tell which atoms have lone pairs of electrons (gray =
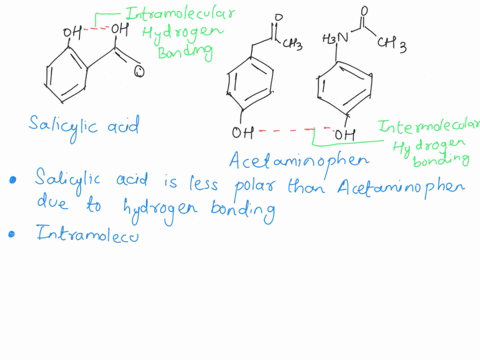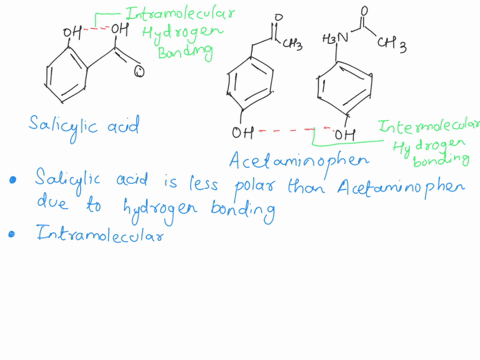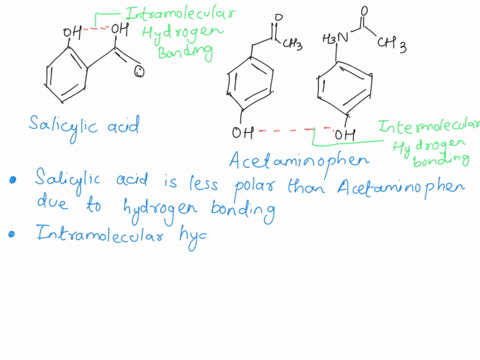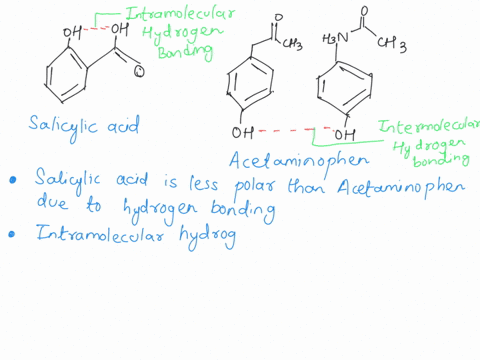
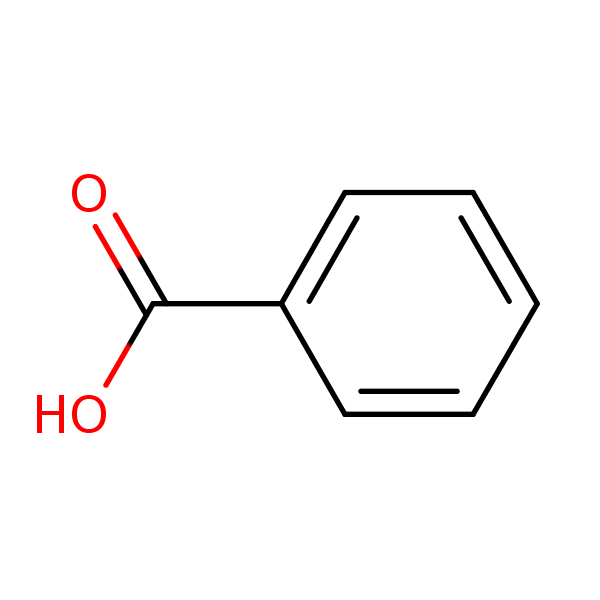
Between acetylsalicylic acid and benzoic acid, which is most polar if you look only at the structure (not at solubility in water g/l), and why? Why is that structure most polar? -
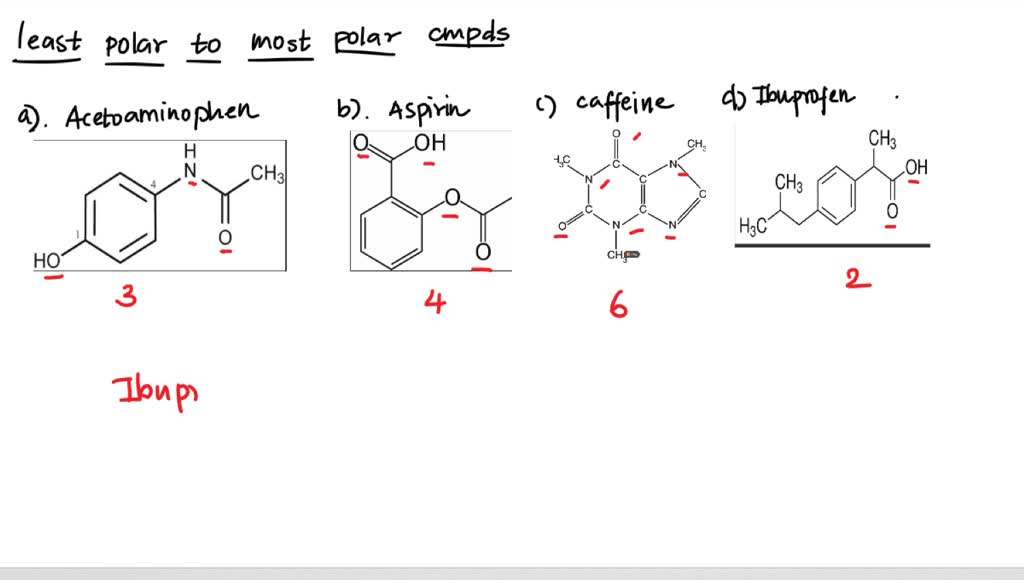
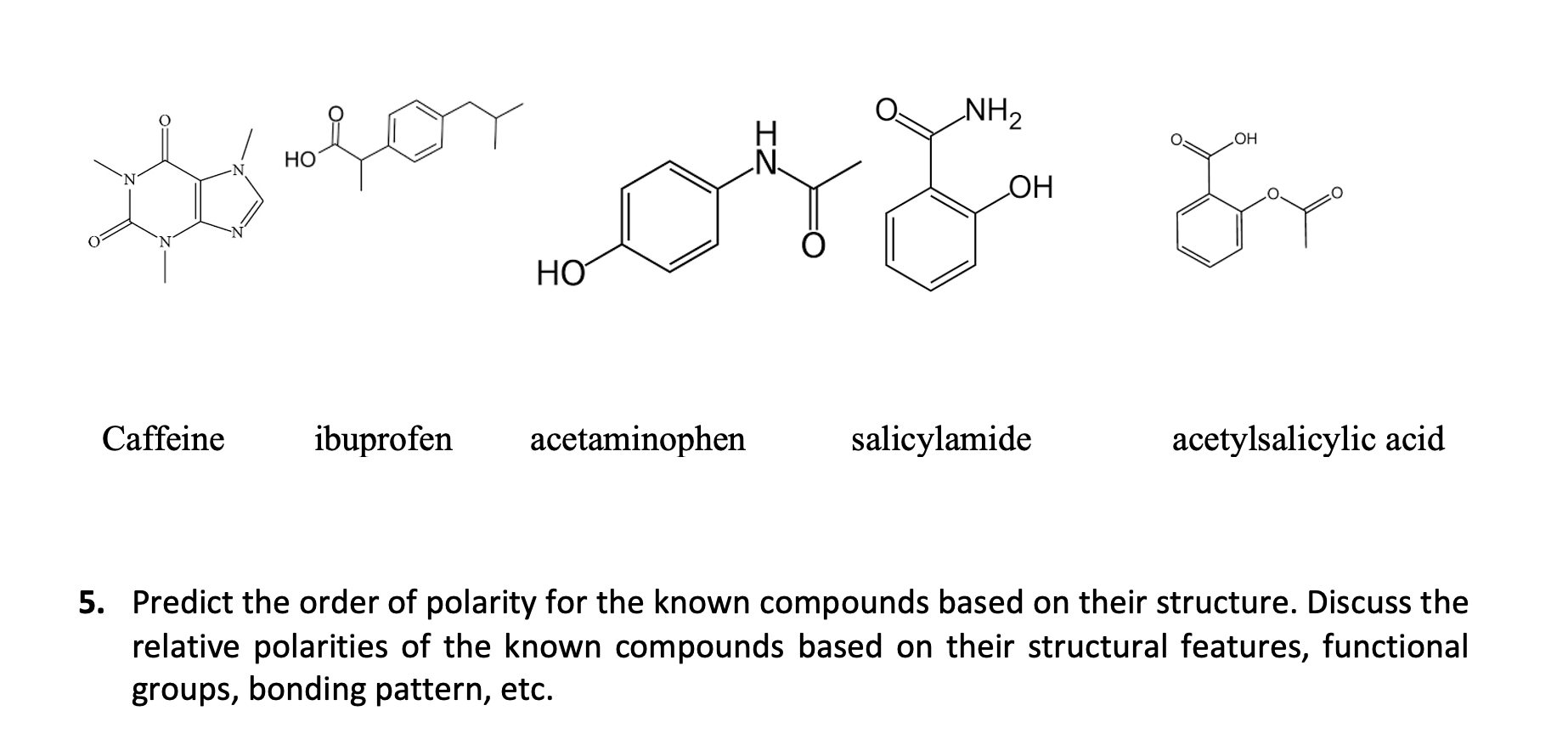
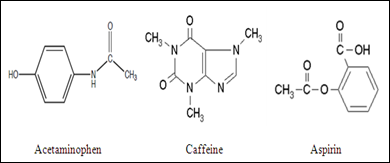
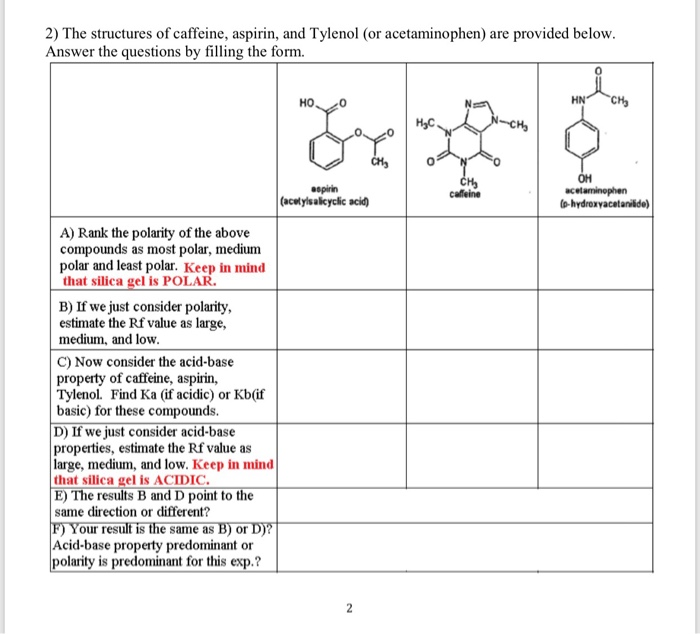
SOLVED: Q2) List the following compounds from least polar to most polar: a. (Acetaminophen, Aspirin, Caffeine, and Ibuprofen) D caffeine > Acetaminophen > phenacetin > Aspirin > ibuprofen Aspirin is more polar

Which compound do you expect to be most polar: aspirin, acetaminophen, or caffeine? Explain. | Homework.Study.com